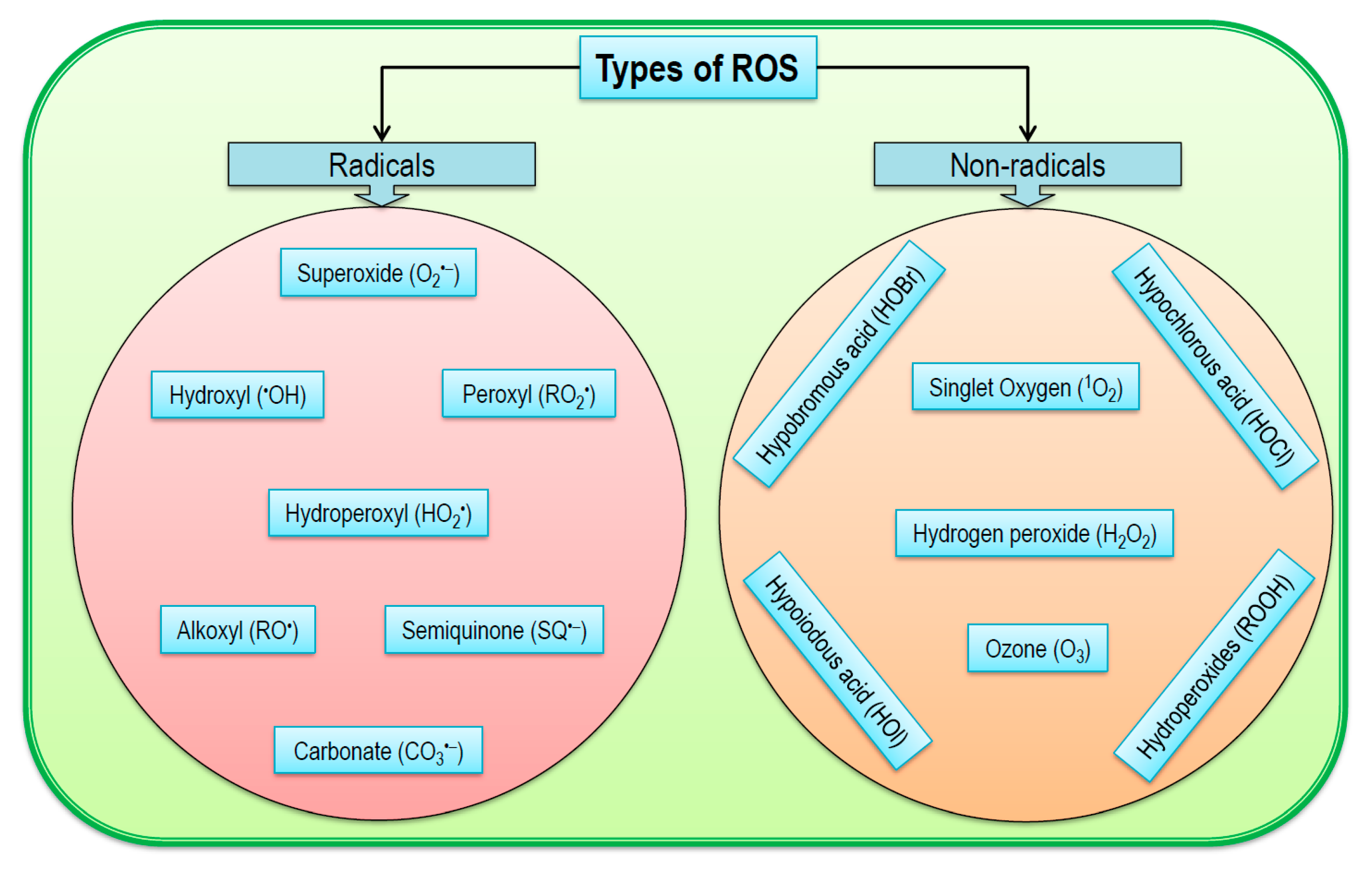
In ROS biology and medicine, the target molecules usually include proteins, lipids, and nucleic acids, among others. The term antioxidant can be defined literally as any substance that can prevent, reduce, or repair the ROS-induced damage of a target biomolecule. In addition, a number of components derived from dietary sources also possess antioxidant activities in biological systems. Mammals including humans have evolved a series of antioxidant defenses to protect vital biomolecules from ROS and related species-mediated damage. In this context, the endogenously formed electrophilic 8-nitroguanosine 3′,5′-cyclic monophosphate acts as a second messenger for nitric oxide-mediated signal transduction (also see Section 5). While high levels of electrophiles, especially α,β-unsaturated aldehydes, cause overt cell and tissue injury, these electrophilic species at non-cytotoxic levels can interfere with cell signaling. Indeed, formation of electrophiles is a major mechanism of chemical toxicity. The term xenobiotic can be defined as any substance that does not occur naturally in the human body. The term electrophile refers to an electron-deficient chemical species that undergoes covalent reactions by accepting an electron pair from an electron-rich biomolecule (also known as a nucleophile).Įlectrophilic species can also be derived from biotransformation of xenobiotics. These include α,β-unsaturated aldehydes, ω-6 and ω-3 unsaturated fatty acids, as well as nitro-fatty acids. Reactions of these reactive species with biomolecules also generate a large array of secondary electrophilic products (also known as electrophiles). ROS are reactive species capable of causing damage to biomolecules, including proteins, lipids, and nucleic acids, leading to cell and tissue injury. Thus, in ROS biology and medicine, the term singlet oxygen, if not specified, usually refers to the 1Δ g state. Due to its highly unstable nature, ( 1Σ g+) 1O2 is generally regarded to lack any biological significance. ( 1Σ g+) 1O 2 is a free radical because it contains two unpaired electrons, whereas ( 1Δ g) 1O 2 is a non-radical. As illustrated in Figure 1, singlet oxygen can exist in two states: the delta state ( 1Δ g) and sigma state ( 1Σ g+). Examples of the non-radical ROS include hydrogen peroxide, peroxynitrite, hypochlorous acid, and ozone. On the other hand, several ROS do not contain any unpaired electrons, and as such, they are not free radicals. Examples of oxygen radicals include superoxide, hydroxyl, peroxyl, and alkoxyl radicals. An unpaired electron refers to the one that occupies an atomic or molecular orbital by itself. The term free radical is defined as any chemical species capable of independent existence that contains one or more unpaired electrons. They are hence also called oxygen radicals or oxygen free radicals. Among the ROS listed above in Section 2.1, some contain unpaired electrons, and thus belong to free radicals.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
